Researchers from UC Riverside, and Rowan University have developed a bioengineered patch that generates its own oxygen supply, tackling one of tissue engineering’s oldest unsolved problems.
The study also saw contributions from UC Berkeley, Tarleton State University in Texas, the Terasaki Institute for Biomedical Innovation in Los Angeles.
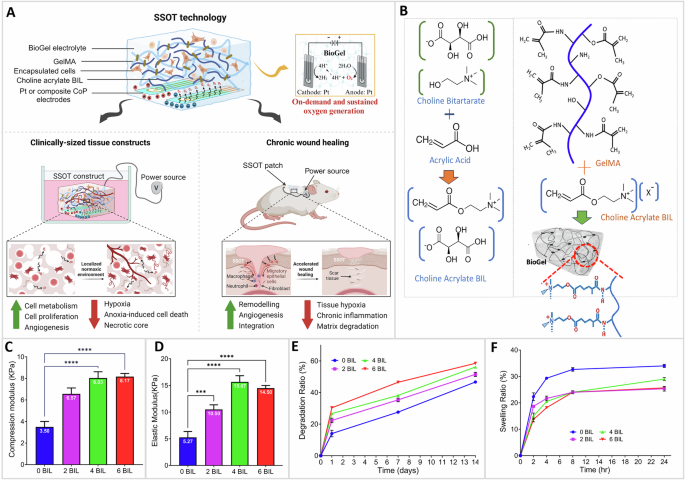
Published in Communications Materials, the work describes a platform the team calls a smart self-oxygenating tissue system (SSOT). It works by running a small electric current through a specially designed gel that contains water, splitting water molecules into oxygen and hydrogen through electrolysis. The oxygen is released directly into the surrounding tissue, on demand, without relying on the bloodstream.
That matters because cells can only survive if a blood vessel is within about 100 to 200 μm of them, roughly the width of two human hairs. In thin tissue samples, oxygen diffuses through naturally. But building tissue thick enough to be clinically useful, the kind that could one day substitute for a damaged organ or repair a large wound, means cells in the interior are too far from any vessel to survive.
Every approach tried so far, including oxygen-carrying molecules, porous scaffolds, and growth factors that stimulate new vessel formation, has failed to sustain cells reliably through that critical window before new vasculature establishes itself.
Engineering Oxygen From Within
The SSOT sidesteps this by generating oxygen inside the scaffold itself. The gel combines GelMA and a bio-ionic liquid made from choline, a nutrient the body naturally produces. Together they form a conductive, adhesive hydrogel the team calls BioGel, which acts as both the electrolyte that carries the current and the environment in which cells live and grow.
As detailed in the study, the electrode that drives the reaction comes in two versions. One uses platinum, a reliable and well-studied material for this kind of electrochemical work. The other uses a composite electrode made from cobalt phosphate, a clay-like mineral called Laponite, and GelMA, formulated specifically so it can be 3D printed into custom shapes using an extrusion bioprinter.
That printability could eventually allow clinicians to tailor electrode geometry to the specific contours of a patient’s wound or implant site.
The choline-derived bio-ionic liquid contributes more than conductivity. Adding it at a 4% concentration more than doubled the gel’s stiffness compared to plain GelMA, and it dramatically slowed enzymatic breakdown. Plain GelMA lost more than 90% of its mass to a collagen-digesting enzyme within three days, and the BioGel formulation lost only about 25% over a full week under the same conditions.
Molecular simulations helped explain why the bio-ionic liquid also improves oxygen delivery: the choline molecules reduce how strongly oxygen sticks to the platinum surface, pushing more of it outward into the tissue.
Putting the Platform to the Test
In laboratory cell culture tests, the team embedded three different human cell types in SSOT constructs and exposed them to severe oxygen deprivation, roughly equivalent to conditions deep inside a thick tissue implant.
After two weeks, cell survival rates in the SSOT group ranged from 74 to 79% depending on cell type. Without the oxygen-generating system, survival dropped to between 38 and 46%. The platform also increased secretion of a protein called VEGF, which the body uses to signal the growth of new blood vessels.
The researchers then implanted SSOT constructs under the skin of rats to assess how the body responds to the device. After 28 days, immune reaction was minimal and infiltration of macrophages, the cells that typically signal rejection of a foreign material, was low. That result matters considerably for any device intended for long-term implantation.
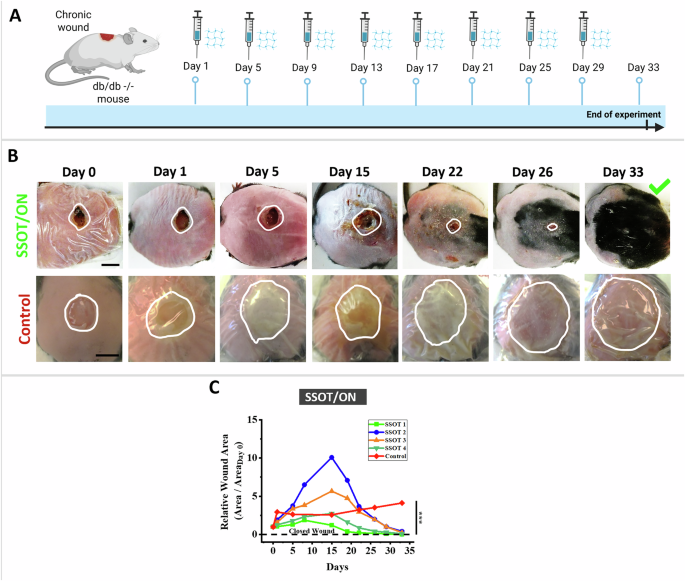
However, the most revealing tests came from animal wound healing trials. The team tested SSOT patches on diabetic mice engineered to develop chronic, non-healing wounds by suppressing two key antioxidant enzymes at the time of injury, allowing bacteria naturally present on the skin to establish persistent infections.
These wounds did not heal on their own and in untreated animals continued to worsen throughout the observation period. Mice treated with battery-powered SSOT patches showed significant wound closure within 33 days, with tissue analysis confirming organized collagen formation and proper skin layer regeneration rather than scar tissue.
Whether the platform can clear the substantial hurdles of human clinical trials remains to be seen. The system operates at just 1 Volt and maintained stable electrochemical performance over 30 days of on-off cycling, but testing in larger animals and eventually humans lies ahead. As a proof of concept, it presents a technically credible answer to a problem that has constrained tissue engineering for decades.
Titled “A smart self-oxygenating system for localized and sustained oxygen delivery in bioengineered tissue constructs,” the study was conducted by Vaishali Krishnadoss, Baishali Kanjilal, Aihik Banerjee, Prince David Okoro, Mohammad Khavani, Proma Basu, Nourouddin Sharifi, Johnson V. John, Manuela Martins-Green, Amos Mugweru, Mohammad R. K. Mofrad, Arameh Masoumi, and Iman Noshadi.
3D Printing Industry is inviting speakers for its 2026 Additive Manufacturing Applications (AMA) series, covering Energy, Healthcare, Automotive and Mobility, Aerospace, Space and Defense, and Software. Each online event focuses on real production deployments, qualification, and supply chain integration. Practitioners interested in contributing can complete the call for speakers form here.
To stay up to date with the latest 3D printing news, don’t forget to subscribe to the 3D Printing Industry newsletter or follow us on LinkedIn.
Explore the full Future of 3D Printing and Executive Survey series from 3D Printing Industry, featuring perspectives from CEOs, engineers, and industry leaders on the industrialization of additive manufacturing, 3D printing industry trends 2026, qualification, supply chains, and additive manufacturing industry analysis.
Featured image shows characterization of the SSOT technology and its function in tissue engineering. Image via Vaishali Krishnadoss et al., Communications Materials.