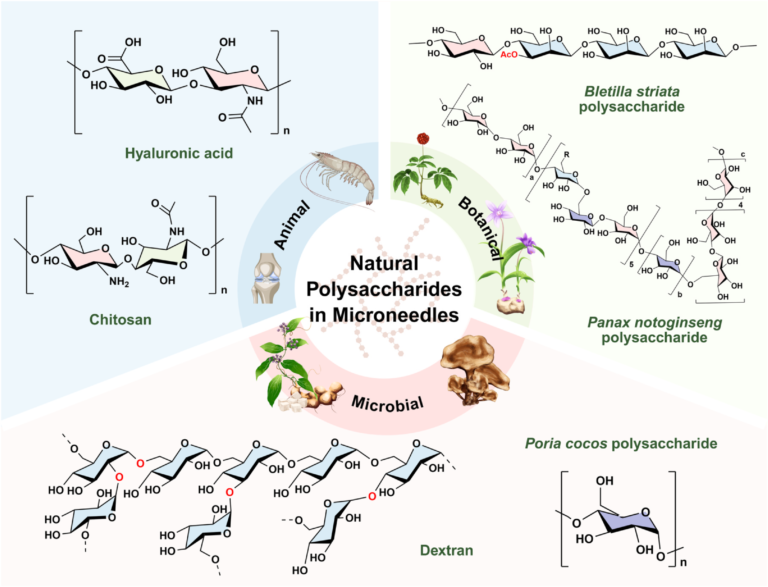
Researchers have reviewed the use of polysaccharide-based microneedles (PMNs) for cancer immunotherapy, examining how additive manufacturing methods, including 3D printing, can support the development of transdermal drug delivery systems. The study, published in Glycoscience & Therapy, focuses on the role of natural polymer materials in enabling minimally invasive treatment approaches that combine drug delivery with immune system activation.
Microneedles are micro-scale structures designed to penetrate the outer layer of the skin and create temporary channels for therapeutic delivery. According to the authors, this approach allows drugs to bypass gastrointestinal degradation while targeting immune-active regions of the skin. Polysaccharides such as hyaluronic acid, chitosan, and alginate are highlighted as key materials due to their biocompatibility, biodegradability, and ability to support controlled drug release.

Polysaccharide microneedles for localized and controlled therapy
The reviewed systems are designed to deliver a range of therapeutic agents, including small-molecule drugs, proteins, and nanoparticle-based formulations. In cancer applications, these microneedles can be used to administer immunotherapeutic compounds directly to the tumor microenvironment or surrounding tissue. The authors note that localized delivery may reduce systemic side effects while improving treatment efficiency.
Several microneedle configurations are discussed, including dissolvable and hydrogel-based structures capable of releasing drugs over time. Functional designs incorporating pH-responsive and enzyme-responsive materials are also examined, enabling drug release triggered by conditions specific to tumor environments. In addition, electrically responsive systems are explored as a method for externally controlled drug delivery.


Additive manufacturing enables microneedle design flexibility
The study identifies additive manufacturing as a potential method for fabricating microneedle arrays with complex geometries and customized designs. Compared to conventional molding techniques, 3D printing can enable rapid prototyping and structural optimization of microneedle architectures.Vat photopolymerization processes such as stereolithography (SLA) and digital light processing (DLP) are typically used for microneedle fabrication due to their high resolution, though their compatibility with polysaccharide materials remains limited.
However, the authors highlight challenges associated with directly printing polysaccharide-based materials. These include limited mechanical strength and difficulties in controlling rheological properties during fabrication. As a result, current approaches often combine 3D printing with indirect methods, such as producing molds that are later filled with functional biomaterials.


Manufacturing and clinical translation challenges remain
Despite ongoing research, the authors emphasize that polysaccharide microneedle systems face several limitations. These include constraints on drug loading capacity, variability in natural material properties, and challenges in achieving consistent large-scale manufacturing. Clinical validation is also limited, with most systems still in preclinical or early-stage research.
The review concludes that while polysaccharide microneedles represent a promising platform for cancer immunotherapy, further development is required to address manufacturing reliability, material standardization, and regulatory requirements before widespread clinical adoption can be achieved.
3D printing microneedle fabrication advances highlight material and process constraints
Previous work in additive manufacturing has demonstrated the feasibility of producing microneedle arrays for transdermal drug delivery using high-resolution processes. Researchers have 3D printed hollow microneedles designed for controlled delivery applications, while other studies have focused on optimizing micro-scale printing parameters to improve structural fidelity and reproducibility. These developments show that additive manufacturing can reliably produce the geometries required for microneedle systems, particularly when using vat photopolymerization techniques capable of achieving micron-level resolution.
At the same time, efforts to commercialize 3D printed microneedle technologies, including initiatives supported by Continuity Biosciences and Joseph DeSimone, indicate ongoing interest in scaling these systems beyond laboratory settings. However, existing approaches are primarily focused on producing microneedle structures using high-resolution photopolymer-based processes, with less emphasis on integrating bioactive materials directly into the printing workflow. This highlights a gap between manufacturable architectures and functional biomaterials, which has led to hybrid fabrication strategies where 3D printing defines the structure, and therapeutic materials are incorporated separately.
3D Printing Industry is inviting speakers for its 2026 Additive Manufacturing Applications (AMA) series, covering Energy, Healthcare, Automotive and Mobility, Aerospace, Space and Defense, and Software. Each online event focuses on real production deployments, qualification, and supply chain integration. Practitioners interested in contributing can complete the call for speakers form here.
To stay up to date with the latest 3D printing news, don’t forget to subscribe to the 3D Printing Industry newsletter or follow us on LinkedIn.
Explore the full Future of 3D Printing and Executive Survey series from 3D Printing Industry, featuring perspectives from CEOs, engineers, and industry leaders on the industrialization of additive manufacturing, 3D printing industry trends 2026, qualification, supply chains, and additive manufacturing industry analysis.
Feature image shows structures of representative polysaccharides used in microneedle systems. Image via Zhang et al.