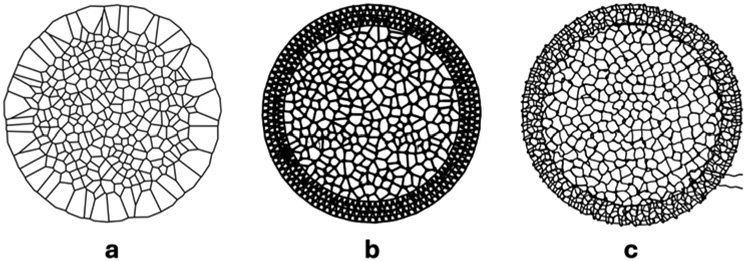
Researchers from Politecnico di Torino and Maastricht University have developed a bioinspired scaffold design approach using a custom Voronoi path generator for extrusion-based 3D printing. Published in Biomaterials Science, the study introduces a Python-based software tool that enables the fabrication of irregular, biomimetic structures through melt electrowriting (MEW) and fused deposition modelling (FDM). The goal is to create more physiologically relevant in vitro lung tissue models.
The system generates continuous toolpaths for complex Voronoi geometries that are difficult to produce using standard slicing software. These geometries were used to create scaffolds that replicate alveolar tissue architecture.The printed structures were combined with an electrospun nanofibrous membrane, forming a multi-scale construct designed to mimic the alveolar-capillary barrier.
Custom toolpath generation for irregular biomimetic structures
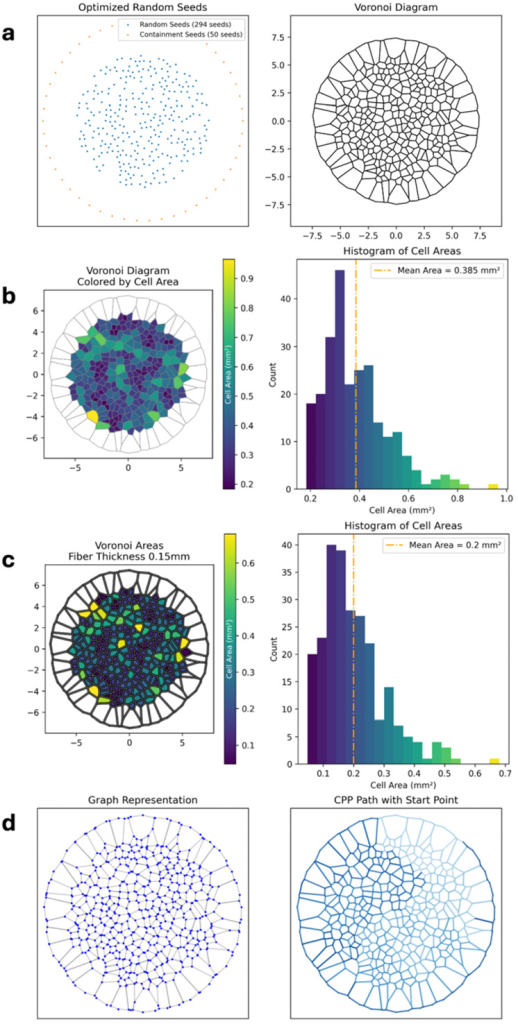
The study highlights that conventional slicing tools are optimized for regular or parametric geometries, lacking the ability to generate continuous extrusion paths for non-repeating structures like Voronoi patterns. To address this limitation, the team developed a custom Python-based software tool called the Voronoi Path Generator (PyVoroGen), which converts Voronoi layouts into G-code optimized for extrusion-based additive manufacturing.
The software allows users to define parameters such as seed number, pattern diameter, and fiber thickness through a graphical user interface. It generates continuous toolpaths using graph theory algorithms, ensuring compatibility with MEW processes that require uninterrupted extrusion. For FDM, additional processing steps are implemented to avoid material overlap by lifting the extruder when segments are revisited to prevent collisions with previously deposited material.
In addition to toolpath generation, the software offers predictive analysis of scaffold properties, including porosity and pore area, establishing a direct link between design parameters and expected print outcomes.
Fabrication using MEW and FDM processes
The Voronoi-based scaffolds were fabricated using both melt electrowriting and fused deposition modelling. Confocal imaging confirmed accurate reproduction of the designed geometries, demonstrating the feasibility of producing non-repeating, biomimetic architectures with extrusion-based 3D printing.
Differences were observed between the two fabrication methods. MEW enabled the production of finer fibers, with an average diameter of approximately 92 µm, while FDM produced thicker fibers and showed higher geometric fidelity in node definition, despite minor connection defects that occasionally led to merging of adjacent cells. MEW structures, by contrast, exhibited reduced fidelity in shorter segments due to rapid directional changes.
Porosity measurements showed close agreement with software predictions, with normalized measured-to-theoretical deviations of approximately 2.2% for FDM and 1.7% for MEW scaffolds.
Hybrid scaffold design integrates electrospinning
Following fabrication, the 3D printed Voronoi backbones were combined with an electrospun membrane composed of polycaprolactone (PCL) and gelatin. This membrane, with an average thickness of approximately 3 µm, was deposited directly onto the printed structures, forming a composite scaffold.
Scanning electron microscopy revealed that the nanofibrous layer conformed to the underlying Voronoi architecture, partially bridging the pores and creating a three-dimensional membrane structure that integrates micro-scale printed fibers with nano-scale electrospun fibers.

In vitro co-culture demonstrates compartmentalized barrier formation
To evaluate biological performance, the scaffolds were used to culture human alveolar epithelial (A549) and endothelial (HUVEC) cells on opposite sides of the membrane. The constructs were maintained under air–liquid interface conditions, with a total culture period of ten days.
Results showed successful cell adhesion and proliferation across both compartments, with epithelial cells forming a continuous layer on the apical side and endothelial cells colonizing the basolateral surface. Immunofluorescence imaging confirmed the formation of a compartmentalized cellular structure resembling the alveolar-capillary barrier.
The thin electrospun membrane enabled simultaneous visualization of both cell layers and supported interaction across the scaffold. The Voronoi architecture also influenced spatial organization, with the printed microfibers affecting cell morphology.

Towards more biomimetic in vitro tissue models
The study indicates that the platform enables the fabrication of scaffolds with increased architectural complexity compared to conventional scaffold geometries. By combining custom computational design, extrusion-based 3D printing, and electrospinning, the approach provides a method for producing more physiologically relevant in vitro models.
While the current scaffolds do not yet match the scale of native alveolar structures, the study demonstrates a framework for integrating biomimetic geometry into additive manufacturing workflows for tissue engineering applications.
Pursuing more realistic scaffold architectures in tissue engineering
Efforts to improve 3D printed scaffolds for biomedical applications have increasingly focused on replicating native tissue structure. Researchers at UNSW Canberra, for example, have developed 3D printed bone scaffolds designed to more closely match natural bone architecture, aiming to improve tissue regeneration outcomes. Similarly, recent work on spinal cord repair has demonstrated that scaffold structure plays a critical role in supporting functional recovery, with 3D printed constructs enabling guided cell growth and tissue integration.
While these approaches highlight the importance of geometry in determining biological performance, most rely on relatively regular or controlled architectures that remain compatible with existing manufacturing workflows. Translating more irregular, heterogeneous designs into printable structures remains a challenge. The method presented in this study addresses this limitation by enabling continuous toolpath generation for non-repeating geometries, supporting the fabrication of scaffold architectures that more closely reflect native tissue organization.
Titled “Bioinspired scaffold design using a custom Voronoi path generator for extrusion-based 3D printing,” the study was conducted by Federico Farina, Michela Licciardello, Lorenzo Moroni, Joanna Babilotte, Gianluca Ciardelli, and Chiara Tonda-Turo.
3D Printing Industry is inviting speakers for its 2026 Additive Manufacturing Applications (AMA) series, covering Energy, Healthcare, Automotive and Mobility, Aerospace, Space and Defense, and Software. Each online event focuses on real production deployments, qualification, and supply chain integration. Practitioners interested in contributing can complete the call for speakers form here.
To stay up to date with the latest 3D printing news, don’t forget to subscribe to the 3D Printing Industry newsletter or follow us on LinkedIn.
Explore the full Future of 3D Printing and Executive Survey series from 3D Printing Industry, featuring perspectives from CEOs, engineers, and industry leaders on the industrialization of additive manufacturing, 3D printing industry trends 2026, qualification, supply chains, and additive manufacturing industry analysis.
Feature image shows theoretical Voronoi architecture and bright-field images of printed structures. Image via Farina et al.